WHAT IS RED YEAST RICE?
Antioxidant flavonoids in red yeast rice:
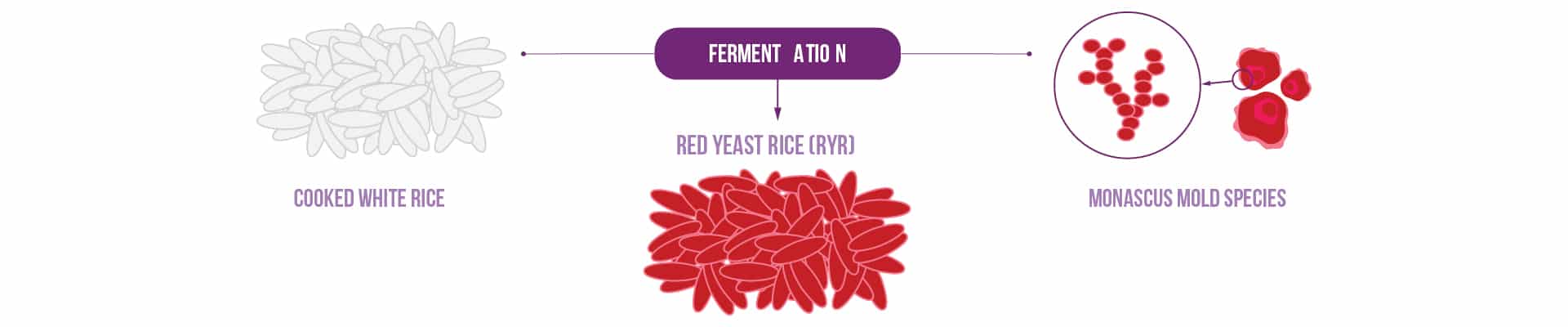
Red yeast rice is a brick-red colored superfood with medicinal properties and a long history. It’s made by fermenting cooked white rice with a species of yeast from the Monascus family, then drying the product and grinding it into a fine powder.
Red yeast rice has been a staple of Traditional Chinese Medicine and a food source for over a thousand years.
In addition to the vibrant color and rich flavor, Monascus adds many beneficial molecules to the rice. One important addition is monacolin K, and it became the first drug ever prescribed to lower blood cholesterol levels.
Monacolin K was the first member of what became a family of drugs called statins. While statins are effective at lowering blood cholesterol, they can also come with side effects.
Many red yeast rice supplements were effective at reducing blood cholesterol due to their high levels of monacolin K, but concerns about safety and side effects led to the banning of these supplements from the market in the USA and Canada.
Meanwhile, scientists noticed that some strains of Monascus did not produce monacolin K but were still able to provide antioxidant benefits. Thus, a search for the active ingredients in Monascus led to the discovery and development of a new type of red yeast rice, called Ankascin 568-R. This form is safe, completely free of monacolin K.
The sale of Ankascin is approved by both Health Canada and the United States Food and Drug Administration (FDA), and its efficacy is being defined by several clinical trials and dozens of ongoing research studies.
THE BENEFITS OF RED YEAST RICE
Many different molecules are produced by Monascus in the fermentation process that produces red yeast rice, and many of these may contribute to the beneficial antioxidant effects of this supplement. These molecules include unsaturated fatty acids, pigments (monascin and ankaflavin are yellow pigments), and phytosterols. Many of these have positive biological activity. For instance, the red pigments monascorubrin and rubropunctatin are antibacterial.
WHO CAN TAKE RED YEAST RICE WITH ANKASCIN?
Ankascin is safe for most people; however, it should be taken with caution under certain conditions.
Consult a healthcare practitioner if you are breastfeeding, taking other medications, if you suffer from liver or kidney disease, if you have received an organ transplant, or for use beyond 8 weeks. If you experience muscle pain, tenderness and/or weakness, discontinue use and consult a healthcare practitioner. Do not use if you are pregnant or plan to become pregnant.
References:
Cohen, P. A., Avula, B. and Khan, I. A. (2017). Variability in strength of red yeast rice supplements purchased from mainstream retailers. European Journal of Preventive Cardiology 24: 1431–1434.
Martínková, L., Patáková-Jůzlová, P., Křen, V., Kučerová, Z., Havlíček, V., Olšovský, P., Hovorka, O., Říhová, B., Veselý, D., Veselá, D., et al. (1999). Biological activities of oligoketide pigments of Monascus purpureus. Food Additives and Contaminants 16: 15–24.
Campoy, S., Sierra, S., Suarez, B., Ramos, M. C., Velasco, J., Burgos, J. S. and Adrio, J. L. (2010). Semisynthesis of novel monacolin J derivatives: hypocholesterolemic and neuroprotective activities. The Journal of Antibiotics 63: 499–505
About the Author
AOR
Let’s keep in touch
Don’t worry we only send updates monthly on offers, product changes, and new opportunities for you.
"*" indicates required fields